Hair loss affects millions of people around the globe, and it greatly influences self-confidence and self-esteem. However, with the invention of PP405, a new topically administered peptide created by Pelage Pharmaceuticals, a brainchild of inventions originating from UCLA. Unlike traditional medications available in the market, such as minoxidil or finasteride, it works by awakening dormant stem cells in hair follicles, which are restored through a new metabolic route. The invention of PP405 works fully by targeting the core problem of hair loss, which ensures that it induces thick and terminal hair growth with few side effects. Thus, we shall examine how and through which mechanisms PP405 may revolutionize hair restoration.
Understanding Hair Loss
Loss of hair is mostly due to genetic, aging, hormonal imbalances (DHT), stress, anxiety, some health issues, and chemotherapy. Currently, the main treatments – Minoxidil and Finasteride only slow down hair loss and stimulate the growth of existing hair. They produce very limited new hair growth and require long-term usage. Consequently, there is a pressing need for more restorative, safer, and widely effective therapies.
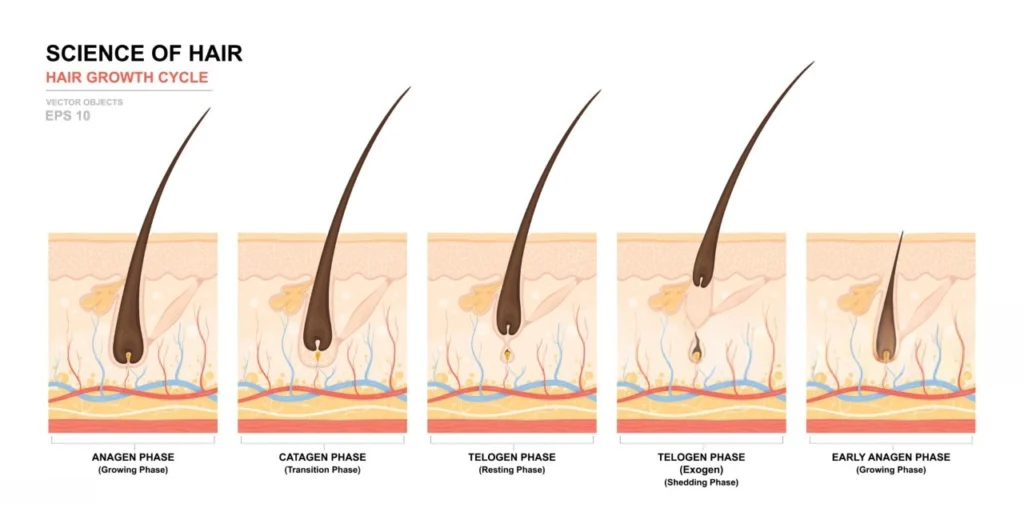
What is the PP405 Hair Peptide?
PP405 is a topical small molecule inhibitor of the mitochondrial pyruvate carrier (MPC) designed to activate dormant hair follicle stem cells. Developed by Pelage Pharmaceuticals based on UCLA research, this peptide targets the metabolic state of the follicle rather than androgen levels. Unlike finasteride, which blocks DHT, PP405 forces stem cells to enter a proliferative state by shifting their energy source to glucose. This chemical signal triggers the transition from the resting (telogen) phase to the active growth (anagen) phase. This metabolic shift is the fundamental driver of its regenerative capacity.
How Does PP405 Trigger Follicle Regrowth?
The activation of hair follicle stem cells (HFSCs) occurs through the forced switch from oxidative phosphorylation to glycolysis within the mitochondria. PP405 blocks the pyruvate carrier, effectively “tricking” the cell into a high-energy metabolic state that mirrors natural growth cycles. Clinical biopsies show increased expression of Ki67, a protein marker strictly associated with cellular proliferation, within 24-48 hours of application (1). This rapid cellular response suggests that the peptide initiates the growth cycle significantly faster than traditional irritants. This direct stimulation of the follicle’s “engine” provides a clear advantage over indirect vasodilators.
Clinical Evidence and Research
Phase 1 Trials– Safety and Biomarker Activation
The 2023 Phase 1 study confirms that patients tolerated PP405 with no measurable systemic absorption, while hair follicle stem cells increased Ki67 activity within days to provide significant proof of concept.
Phase 2a Trial – Fast Visible Results
Mid-2024: A randomized placebo-controlled trial on 78 adults used 0.05% PP405 on a daily basis for 4 weeks, and results were measured for 12 weeks. Main findings:
- 31% of PP405-treated patients experienced a >20% increase in hair density at week 8, whereas none in the placebo group
- Visible regrowth even in follicles that previously lacked hair (“terminal hair,” not simply peach fuzz)
- Primary efficacy and pharmacokinetic endpoints, no systemic drug concentrations were maintained.
These results indicate more rapid and regenerative results compared to conventional therapy.
Supporting Data: Clinical Trial Summary
The table showing important clinical trial information as of July 2025 is given below:
| Trial Phase | Participants | Duration | Key Findings | Next Steps |
| Phase 1 (2023) | Small cohort | 1 week | Significant Ki67 increase, safe, no systemic absorption | Phase 2a initiation |
| Phase 2a (2024) | 78 adults | 4 weeks, 12-week follow-up | 31% men showed >20% hair density increase, new growth in bald areas | Phase 3 planned for 2026 |
Is PP405 Better than Minoxidil?
PP405 demonstrates hair growth in the phase 2a trial, in which 31% seeing >20% increase in the density (2). While minoxidil functions as a vasodilator to increase blood flow, it does not fundamentally alter the metabolic dormant state of the follicle. The following table illustrates the clinical distinctions between these modalities:
| Feature | PP405 Peptide | Topical Minoxidil |
| Primary Mechanism | MPC Inhibition (Metabolic) | Vasodilation (Blood Flow) |
| Stem Cell Activation | Direct | Indirect |
| Systemic Absorption | None Detected | Minimal to Moderate |
| Treatment Duration | 4-Week Cycles | Lifelong Daily Use |
| Side Effect Profile | Localized Redness only | Scalp Irritation, Hypertrichosis |
These differences in biological pathways explain why PP405 is classified as a regenerative therapy rather than a maintenance drug.
The ability of this peptide to reactivate previously dormant follicles, rather than just slowing loss, sets it apart as a regenerative therapy.
Application and Use
It is delivered as a 0.05% topical gel, applied once daily. The 4-week randomized portion is followed by an optional open-label extension to assess long‑term safety and durable effects.
Researchers actively test PP405 on individuals with androgenetic alopecia, and early results indicate potential benefits for telogen effluvium, post-chemotherapy hair loss, and other dormant-follicle conditions. The current trials include both men and women.
Benefits and Potential
- Regrowth from dormant follicles—not just slowing hair shedding
- Rapid onset: cell activation within days; visible density gains in as little as 8 weeks
- Minimal side effects: no systemic absorption, lower irritation versus oral/topical drugs
- Regenerative, not just corrective: could complement existing therapies or hair transplant prep
Risks and Limitations
As of now, all the trials have shown no serious adverse events, and localized irritation or redness is minimal and short-lived. Broad systemic risks (e.g., sexual dysfunction from finasteride) are not likely to occur because systemic exposure isn’t detected.
Although pregnancy, lactation, or scalp infections remain contraindications pending broader safety data. Long-term outcomes beyond 12 weeks await completion of ongoing trials.
Future Outlook
Initially licensed in 2018, PP405 has now reached a significant milestone, as Pelage Pharmaceuticals has announced Phase 3 planning for 2026. Assuming positive results and regulatory clearance, a commercial launch could occur between 2027 and 2030. Google Ventures and several prominent dermatology leaders support this phase because PP405 has the potential to become the first-in-class regenerative peptide therapy for hair regrowth.
In the future, it could be used concurrently with minoxidil, PRP, or laser therapy, providing a synergistic effect for hair restoration interventions.
Conclusion
PP405 is a completely novel and exciting Turning Point in Hair Loss Remedies. Its biggest strength is that rather than highlighting a thinning of the hair, it stimulates inactive hair follicles to produce fast, regeneration-oriented, and very safe results. It is a remedy that is currently in the trial stages, yet it does promise hope to those who have not been fortunate in their past treatments—men as well as women. One must watch out for this in the future, as it may completely change the face of alopecia treatments.
Sources
(1) Clinical biopsies show increased expression of Ki67, a protein marker strictly associated with cellular proliferation, within 24-48 hours of application.
(2) PP405 demonstrates hair growth in the phase 2a trial, in which 31% seeing >20% increase in the density.
FAQs
1. Can PP405 regrow hair on bald spots with no visible follicles?
Yes—the initial observations on some studies have shown growth in areas where there was no hair before, which means the dormant follicles have been activated.
2. Is PP405 safe for women?
Yes. The early trials included both men and women, and no systemic drug absorption was detected, which reduces systemic risk.
3. How quickly might I see improvement?
In some cases, cellular activity increases within days; however, some users show>20% density gains by week 8 after 4 weeks of application.
4. Can PP405 be combined with minoxidil or PRP?
Although the early combination has not yet been formally studied together, the early commentary suggests synergistic potential as combination therapies.
5. When could PP405 be on the market?
Researchers plan to conduct the Phase 3 trial for PP405 in 2026, anticipating regulatory approval, with a potential launch between 2027 and 2030.


